GMP Certified in the Philippines: The Ultimate Guide to Compliance and Benefits

Introduction
Obtaining your business
GMP Certified in the Philippines is not merely about regulations — it’s about safeguarding your customers, expanding your market, and gaining trust. Whether you operate in food, pharmaceuticals, cosmetics, or supplements, GMP certification assists you in adhering to local as well as international quality standards. But what exactly is it to be GMP certified? What’s the process? And how can certification benefit your business? Let’s demystify it.What Does It Mean to Be GMP Certified?
GMP refers to Good Manufacturing Practices. These are globally accepted systems that make sure your products are consistently manufactured and controlled in terms of quality standards.
In plain language, it’s about ensuring that your manufacturing process does not facilitate contamination, confusion, or mistakes — particularly when people’s health is at stake.
To be GMP certified is to say that your company has been inspected and satisfies these conditions. It also indicates that you possess:
- A clean, well-organized factory to manufacture
- Accurate documentation and standard procedures
- Trained personnel who know about hygiene and safety
- Problem detection and correction system
These guidelines are applicable across sectors but are especially crucial in:
- Food manufacturing and processing
- Pharmaceuticals
- Cosmetics and personal care
- Herbal supplements and vitamins
Find out more: Wikipedia – Good Manufacturing Practices
Request A Free Quote
Why GMP Certification Is Important in the Philippines
GMP compliance isn’t only good practice — it’s frequently a regulatory requirement.
In the Philippines, Food and Drug Administration (FDA) calls for GMP observance in specific segments such as food, drugs, and cosmetics. Firms that are not up to par will have their licenses suspended or products recalled.
However, in addition to compliance, GMP certification brings significant business benefits:
- Generates confidence among local and global consumers
- Ensures consistency and minimizes wastage
- Paves the way for export and major retail collaborations
- Brings in investors who are serious about quality systems
Go to the Philippine FDA website for an overview of regulations and guidelines.
Who is required to become GMP certified in the Philippines?
You might be obligated — or highly recommended — to become GMP certified if you’re involved in:
- Food processing: meat, poultry, seafood, dairy, baked foods, etc.
- Pharmaceuticals: OTC drugs, syrups, tablets, injectables
- Cosmetics & toiletries: soaps, lotions, perfumes, skin care
- Herbal & traditional products
- Supplements: vitamins, minerals, powdered mixes
- Third-party or contract manufacturers
- Packaging companies handling food or pharma
Even small and medium-sized businesses (SMEs) are opting for certification to remain competitive, particularly if they intend to export or get into chain-store distribution.
Step-by-Step Process to Get GMP Certified
GMP certification can sound technical, but broken into steps, it’s a straightforward and easy process.
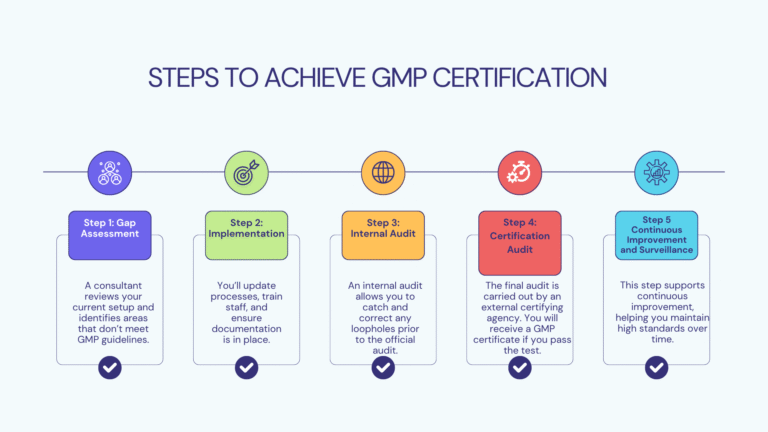
Benefits of Being GMP Certified
Certification means so much more than a neat picture on the wall of your office.
Here’s the reality of what it actually does for your company:
- Enhanced product safety and quality
- Reduced risk of recalls or contamination
- Increased market confidence and consumer trust
- Qualification for local FDA licensing
- Seamless export processing to the US, EU, and ASEAN
- Edge over non-certified competitors
Whether you are a new food processor or an existing cosmetic laboratory, GMP provides the credibility necessary to broaden your scope.
Common Challenges in Getting GMP Certified
It’s not always smooth sailing. There are roadblocks like:
- Insufficient quality documentation or control of the process
- Poor facility layout (e.g., inefficient material or staff flow)
- Workers not adhering to hygiene practices
- Misinterpretation of what the auditors are looking for
- Budget limitations
Having a compliance partner — such as Maxicert — can resolve such troubles and make your audit worry-free.
Cost of GMP Certification in the Philippines
There is no fixed cost, but fees are based on:
- Facility size
- Product type (food, drug, cosmetic)
- Location number
- Audit readiness (new vs. mature system)
Typically, the cost covers:
- Pre-audit gap analysis
- Documentation support
- Audit cost
- Corrective action assistance
While the initial investment might be high, the long-term ROI — in reputation, risk reduction, and growth — is well worth it.
Why Work with Maxicert for Your GMP Certification?
We’ve guided companies throughout the Philippines to achieve GMP certification with ease and confidence.
With Maxicert, you receive:
- Document-to-audit consultation on a step-by-step basis
- Domestic knowledge with international standards
- Quick turnaround with tailored solutions
- Transparent pricing with no additional fees
- ISO, HACCP, and other integrated system support
Whether you’re new to compliance or simply want to renew your certificate, we make it easy, quick, and budget-friendly.
Conclusion
The world demands safer, higher-quality products than ever before. Consumers demand it. Regulators are enforcing it. And foreign markets are more rigorous than ever.
Get ahead of the trend. Get certified. Contact us today.
Becoming GMP certified is a step in the right direction. It enables you to produce improved products, minimize risk, and venture into new markets with confidence.
- Initial consultation
- Expedited documentation
- Local assistance with worldwide experience
Reach out to us now to book an appointment and obtain your GMP certification hassle-free.

Get In Touch

Get In Touch

Get In Touch
Need A Free Estimate?
Get a free consultation and Checklist to get certified for ISO , HALAL, CE Mark Certification.
FAQ
What does it mean to be GMP certified?
GMP certification means your business follows Good Manufacturing Practices to ensure product safety, hygiene, and quality during production.
Who needs GMP certification in the Philippines?
Companies in food, pharmaceuticals, cosmetics, and supplements need GMP certification to meet regulatory standards and build consumer trust.
How long does GMP certification take?
The process typically takes 4 to 12 weeks, depending on your company’s readiness, documentation, and size of operations.
Is GMP certification mandatory in the Philippines?
Yes, for many industries regulated by the FDA Philippines—like food and drug manufacturing—GMP compliance is legally required.



